問題詳情
76. The boiling points of halogens (see table right) increases from F2to I2. This is the result of an increase in which quantity fromF2to I2? 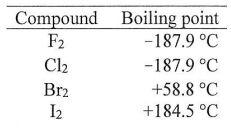
(A) Ionic bonding
(B) Covalent bond strength
(C) Electron affinity
(D) Van der Waals forces
(E) Nuclear quadrupole moment
參考答案
答案:D
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增