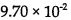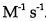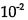問題詳情
27.The reaction: 2 HI→ H2 + I2, is second order and the rate constant at 800 K is
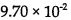
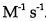
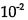
(A) 0.619s
(B) 124 s
(C) 387s
(D) 429 s
參考答案
答案:C
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增
27.The reaction: 2 HI→ H2 + I2, is second order and the rate constant at 800 K is