問題詳情
22. Consider the following reaction at 25°C: 2NO2(g)⇌N2O4(g). The values of ∆Ηο and ∆Sο are -58.03 kJ/mol and-176.6 J/K•mol, respectively. Assuming ∆Ηο and ∆Sο are temperature independent, estimate the value of K at 100.0°C.
(A) 3.00 × 106
(B)
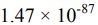
(C) 8.72
(D)
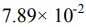
(E)
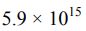
參考答案
答案:D
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增