問題詳情
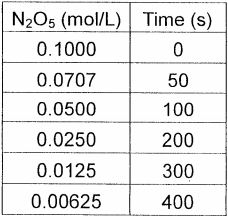
4.The decomposition of N205 in the gas phase wasstudied at constant temperature:2 N O 5(G) → 4 NO2(g) + O2(g)the following results were collected:Using these data, verify that the rate law is first order in [N2O5], and calculate the rate constant.
參考答案
答案:A
難度:簡單0.870899
統計:A(1221),B(74),C(33),D(74),E(0)
難度:簡單0.870899
統計:A(1221),B(74),C(33),D(74),E(0)
用户評論
【Chih Hui】評論
A用鈞