問題詳情
32. 試求AgSCN(S)在 0.003 M NH3水溶液中的溶解度為何?[已知AgSCN的Ksp=1.0×10-12, 且
(A) 1.05×10-5 M
(B) 1.23×10-5 M
(C) 2.10×10-5 M
(D) 2.85×10-5 M
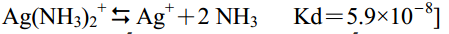
(A) 1.05×10-5 M
(B) 1.23×10-5 M
(C) 2.10×10-5 M
(D) 2.85×10-5 M
參考答案
答案:B
難度:適中0.428571
統計:A(0),B(6),C(4),D(0),E(0)
難度:適中0.428571
統計:A(0),B(6),C(4),D(0),E(0)
用户評論
【去屋尼】評論
let 解離度為 x ,平衡時[Ag+] = y 質量守恆 [Ag+] + [Ag(NH3)2+] = x ,則 [Ag(NH3)2+] = x - yKsp = [Ag+] [SCN-] = y x = 10-12Kd = [Ag+] [NH3]2 / [Ag(NH3)2+] = y (0.003)2 / (x-y) = 5.9 x 10-8Ksp/Kd = x(x-y) / (0.003)2 =( x2-xy)/ (0.003)2 =( x2 - Ksp )/(0.003)2x2= [(0.003)2 x (Ksp / Kd)] + Ksp x = {[(0.003)2 x 10-12 / 5.9 x 10-8 ] + 10-12}1/2 = 1.24 x 10-...