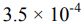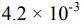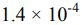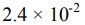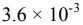問題詳情
18. A 0.15 M solution of a weak acid is 3.0% dissociated. The Ka of the acid is calculated to be _______ .
(A)
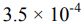
(B)
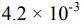
(C)
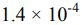
(D)
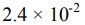
(E)
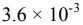
參考答案
答案:C
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增
18. A 0.15 M solution of a weak acid is 3.0% dissociated. The Ka of the acid is calculated to be _______ .
(A)