問題詳情
6. The Ostwald process for the commercial production of nitric acid from ammonia and oxygen involves the following steps:
4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g)
2NO(g) + O2(g) → 2NO2(g)
3NO2(g) + H2O(l) → 2HNO3(aq) + NO(g). The values of
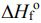
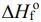
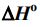
(A) -908 kJ
(B) -112 kJ
(C) -140 kJ
(D) -826 kJ
(E) -220 kJ
參考答案
答案:C
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增