問題詳情
29. At room temperature, a 0.02 mol/L MOH solution was added dropwisely to a 100mL of 0.01 mol/L HA solution, and the curve shown in the figure below indicatesthe pH change of the mixed solution. Which of the following statements is correct?
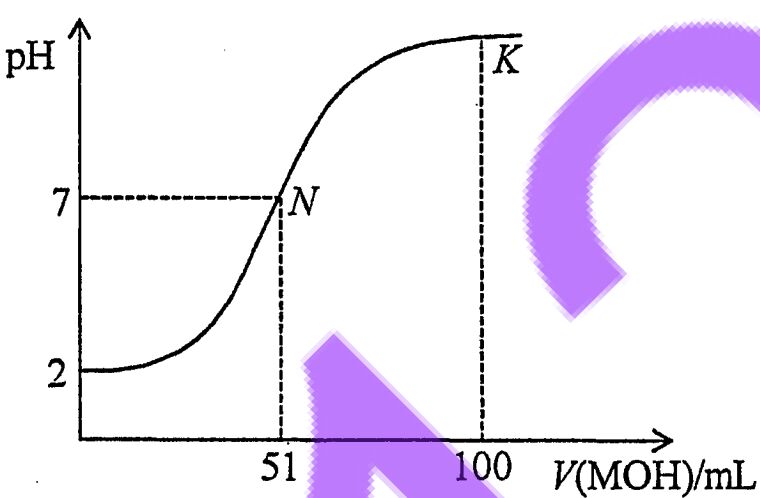
(A)N is the equivalence point.
(B)At N, [A-] =[M+] + [MOH].
(C)At K, [M+] >[A-]> [OH-] >[H+].
(D)HA is a weak acid and MOH is a strong base.
(E) At pH = 7, both HA and MOH are completely consumed.
參考答案
答案:C
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增