問題詳情
15. The first-order isomerization reaction: cyclopropane→ propene, has a rate constant of 1.10
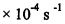
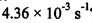
(A)126℃
(B)411℃
(C) 510℃
(D) 540℃
參考答案
答案:D
難度:計算中-1
書單:沒有書單,新增
難度:計算中-1
書單:沒有書單,新增
用户評論
【面試倒楣鬼-22幸運上後西】評論
Rate = aAe(-Ea/RT)則 ☆☆☆☆☆ = 1.1*...
15. The first-order isomerization reaction: cyclopropane→ propene, has a rate constant of 1.10
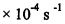
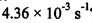
【面試倒楣鬼-22幸運上後西】評論
Rate = aAe(-Ea/RT)則 ☆☆☆☆☆ = 1.1*...